Medical Device Testing Services Market Size, Share, and Trends Analysis Report
CAGR :

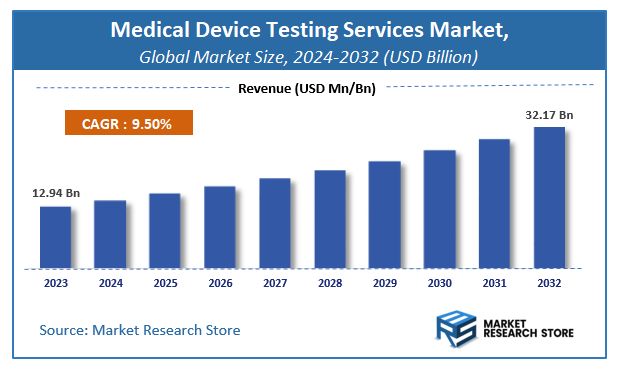
| Market Size 2023 (Base Year) | USD 12.94 Billion |
| Market Size 2032 (Forecast Year) | USD 32.17 Billion |
| CAGR | 9.5% |
| Forecast Period | 2024 - 2032 |
| Historical Period | 2018 - 2023 |
Medical Device Testing Services Market Insights
A latest report by Market Research Store estimates that the Global Medical Device Testing Services Market was valued at USD 12.94 Billion in 2023 and is expected to reach USD 32.17 Billion by 2032, with a CAGR of 9.5% during the forecast period 2024-2032. The report Medical Device Testing Services Market overview, growth factors, restraints, opportunities, segmentation, key developments, competitive landscape, consumer insights, and market growth forecast in terms of value or volume. These structured details offer an all-inclusive market overview, providing valuable insights for investment decisions, business decisions, strategic planning, and competitive analysis.
To Get more Insights, Request a Free Sample
Medical Device Testing Services Market: Overview
The growth of the medical device testing services market is fueled by rising global demand across various industries and applications. The report highlights lucrative opportunities, analyzing cost structures, key segments, emerging trends, regional dynamics, and advancements by leading players to provide comprehensive market insights. The medical device testing services market report offers a detailed industry analysis from 2024 to 2032, combining quantitative and qualitative insights. It examines key factors such as pricing, market penetration, GDP impact, industry dynamics, major players, consumer behavior, and socio-economic conditions. Structured into multiple sections, the report provides a comprehensive perspective on the market from all angles.
Key sections of the medical device testing services market report include market segments, outlook, competitive landscape, and company profiles. Market Segments offer in-depth details based on Services, Phase, and other relevant classifications to support strategic marketing initiatives. Market Outlook thoroughly analyzes market trends, growth drivers, restraints, opportunities, challenges, Porter’s Five Forces framework, macroeconomic factors, value chain analysis, and pricing trends shaping the market now and in the future. The Competitive Landscape and Company Profiles section highlights major players, their strategies, and market positioning to guide investment and business decisions. The report also identifies innovation trends, new business opportunities, and investment prospects for the forecast period.
Key Highlights:
- As per the analysis shared by our research analyst, the global medical device testing services market is estimated to grow annually at a CAGR of around 9.5% over the forecast period (2024-2032).
- In terms of revenue, the global medical device testing services market size was valued at around USD 12.94 Billion in 2023 and is projected to reach USD 32.17 Billion by 2032.
- The market is projected to grow at a significant rate due to rising regulatory compliance requirements and growing medical device innovation.
- Based on the Services, the Biocompatibility Tests segment is growing at a high rate and will continue to dominate the global market as per industry projections.
- On the basis of Phase, the Preclinical and Clinical segment is anticipated to command the largest market share.
- Based on region, North America is projected to dominate the global market during the forecast period.
Medical Device Testing Services Market: Report Scope
This report thoroughly analyzes the medical device testing services market, exploring its historical trends, current state, and future projections. The market estimates presented result from a robust research methodology, incorporating primary research, secondary sources, and expert opinions. These estimates are influenced by the prevailing market dynamics as well as key economic, social, and political factors. Furthermore, the report considers the impact of regulations, government expenditures, and advancements in research and development on the market. Both positive and negative shifts are evaluated to ensure a comprehensive and accurate market outlook.
| Report Attributes | Report Details |
|---|---|
| Report Name | Medical Device Testing Services Market |
| Market Size in 2023 | USD 12.94 Billion |
| Market Forecast in 2032 | USD 32.17 Billion |
| Growth Rate | CAGR of 9.5% |
| Number of Pages | 164 |
| Key Companies Covered | NORTH AMERICAN SCIENCE ASSOCIATES INC., Toxikon Inc., Eurofins Scientific LLC, WuXi AppTec, Charles River Laboratories International Inc., Intertek Group Plc, SGS SA, Sterigenics International LLC, Pace Analytical Services |
| Segments Covered | By Services, By Phase, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Base Year | 2023 |
| Historical Year | 2018 to 2023 |
| Forecast Year | 2024 to 2032 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Medical Device Testing Services Market: Dynamics
Key Growth Drivers
The Medical Device Testing Services market is experiencing significant growth primarily driven by the stringent regulatory landscape governing the development and commercialization of medical devices worldwide. Regulatory bodies such as the FDA in the United States and the MDR in Europe mandate rigorous testing and validation to ensure the safety and efficacy of medical devices before they can be marketed. The increasing complexity of modern medical devices, incorporating advanced electronics, software, and materials, necessitates specialized testing services to verify their functionality and reliability. Furthermore, the growing focus on patient safety and the potential for serious harm from malfunctioning devices underscore the critical need for comprehensive testing. The rising number of medical device companies, including startups and established players developing innovative products, also contributes to the demand for independent testing services.
Restraints
Despite the strong growth drivers, the Medical Device Testing Services market faces certain restraints. The high cost associated with comprehensive medical device testing, particularly for complex devices requiring specialized equipment and expertise, can be a barrier for smaller manufacturers or startups with limited financial resources. The time-consuming nature of thorough testing processes can potentially delay product launches and time-to-market, which is a critical factor in the competitive medical device industry. The availability of in-house testing capabilities within some larger medical device companies might reduce their reliance on external testing services. Furthermore, inconsistencies in regulatory requirements and testing standards across different geographical regions can create complexities and increase costs for manufacturers seeking global market access. The need for specialized expertise and the potential shortage of qualified testing personnel can also pose a restraint on the growth of testing service providers.
Opportunities
The Medical Device Testing Services market presents numerous opportunities for expansion and specialization. The increasing adoption of connected medical devices and the growing focus on cybersecurity within healthcare create a demand for specialized testing services to ensure data security and device integrity. The rising trend of outsourcing non-core activities, including testing, by medical device companies allows them to focus on their core competencies and can drive demand for independent testing providers. The development of new testing methodologies and technologies, such as simulation and digital twins, can enhance the efficiency and comprehensiveness of testing services. The growing emphasis on biocompatibility and sterilization testing for medical devices offers niche opportunities for specialized testing labs. Furthermore, the increasing focus on post-market surveillance and the need for ongoing testing to ensure the continued safety and effectiveness of devices already in use provide a recurring revenue stream for testing services. The expansion of the medical device market in emerging economies with evolving regulatory frameworks also presents significant growth potential for testing service providers.
Challenges
The Medical Device Testing Services market faces several challenges that need to be addressed for sustained growth and to ensure patient safety. Keeping pace with the rapidly evolving technologies and increasing complexity of medical devices requires continuous investment in new testing equipment, methodologies, and expertise. Ensuring compliance with the constantly changing and often stringent regulatory requirements across different regions demands ongoing monitoring and adaptation of testing processes. Maintaining the accuracy, reliability, and impartiality of testing results is crucial for building trust among manufacturers and regulatory bodies. The need for efficient turnaround times without compromising the thoroughness of testing poses a logistical challenge for testing service providers. Furthermore, managing the increasing volume of testing requests while maintaining high quality standards requires scalability and efficient resource management. The potential for misinterpretation of testing standards or regulatory guidelines can lead to delays and disputes. Adapting to the growing demand for more sustainable and environmentally friendly testing practices also presents a challenge for the industry.
Medical Device Testing Services Market: Segmentation Insights
The global medical device testing services market is segmented based on Services, Phase, and Region. All the segments of the medical device testing services market have been analyzed based on present & future trends and the market is estimated from 2024 to 2032.
Based on Services, the global medical device testing services market is divided into Biocompatibility Tests, Chemistry Test and Microbiology & Sterility Test.
On the basis of Phase, the global medical device testing services market is bifurcated into Preclinical and Clinical.
Medical Device Testing Services Market: Regional Insights
The Medical Device Testing Services Market is experiencing robust growth, with North America dominating the global landscape. This leadership is driven by stringent regulatory requirements (FDA, Health Canada), a high concentration of medical device manufacturers, and increasing demand for compliance and safety testing in the U.S. and Canada. North America benefits from advanced healthcare infrastructure, significant R&D investments, and a strong focus on quality assurance for Class II and III devices. Europe follows closely, supported by the EU’s Medical Device Regulation (MDR) and a thriving medtech industry in Germany, France, and the UK. Meanwhile, the Asia-Pacific region is projected to grow at the fastest CAGR, fueled by expanding medical device production, outsourcing trends, and regulatory harmonization in China, India, and Japan. However, North America retains the largest market share (over 40%), owing to its rigorous pre- and post-market testing protocols, cybersecurity testing for connected devices, and dominance in innovative sectors like robotics and AI-driven diagnostics. The region’s leadership is further reinforced by accredited labs and collaborations between test service providers and academic institutions. With global medical device sales rising, North America’s testing services market is poised to maintain its lead through 2030.
Medical Device Testing Services Market: Competitive Landscape
The medical device testing services market report offers a thorough analysis of both established and emerging players within the market. It includes a detailed list of key companies, categorized based on the types of products they offer and other relevant factors. The report also highlights the market entry year for each player, providing further context for the research analysis.
The "Global Medical Device Testing Services Market" study offers valuable insights, focusing on the global market landscape, with an emphasis on major industry players such as;
- NORTH AMERICAN SCIENCE ASSOCIATES INC.
- Toxikon Inc.
- Eurofins Scientific LLC
- WuXi AppTec
- Charles River Laboratories International Inc.
- Intertek Group Plc
- SGS SA
- Sterigenics International LLC
- Pace Analytical Services
The Global Medical Device Testing Services Market is Segmented as Follows:
By Services
- Biocompatibility Tests
- Chemistry Test and Microbiology & Sterility Test
By Phase
- Preclinical and Clinical
By Region
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Market Evolution
This section evaluates the market position of the product or service by examining its development pathway and competitive dynamics. It provides a detailed overview of the product's growth stages, including the early (historical) phase, the mid-stage, and anticipated future advancements influenced by innovation and emerging technologies.
Porter’s Analysis
Porter’s Five Forces framework offers a strategic lens for assessing competitor behavior and the positioning of key players in the medical device testing services industry. This section explores the external factors shaping competitive dynamics and influencing market strategies in the years ahead. The analysis focuses on five critical forces:
- Competitive Rivalry
- Threat of New Entrants
- Threat of Substitutes
- Supplier Bargaining Power
- Buyer Bargaining Power
Value Chain & Market Attractiveness Analysis
The value chain analysis helps businesses optimize operations by mapping the product flow from suppliers to end consumers, identifying opportunities to streamline processes and gain a competitive edge. Segment-wise market attractiveness analysis evaluates key dimensions like product categories, demographics, and regions, assessing growth potential, market size, and profitability. This enables businesses to focus resources on high-potential segments for better ROI and long-term value.
PESTEL Analysis
PESTEL analysis is a powerful tool in market research reports that enhances market understanding by systematically examining the external macro-environmental factors influencing a business or industry. The acronym stands for Political, Economic, Social, Technological, Environmental, and Legal factors. By evaluating these dimensions, PESTEL analysis provides a comprehensive overview of the broader context within which a market operates, helping businesses identify potential opportunities and threats.
- Political factors assess government policies, stability, trade regulations, and political risks that could impact market operations.
- Economic factors examine variables like inflation, exchange rates, economic growth, and consumer spending power to determine market viability.
- Social factors explore cultural trends, demographics, and lifestyle changes that shape consumer behavior and preferences.
- Technological factors evaluate innovation, R&D, and technological advancements affecting product development and operational efficiencies.
- Environmental factors focus on sustainability, climate change impacts, and eco-friendly practices shaping market trends.
- Legal factors address compliance requirements, industry regulations, and intellectual property laws impacting market entry and operations.
Import-Export Analysis & Pricing Analysis
An import-export analysis is vital for market research, revealing global trade dynamics, trends, and opportunities. It examines trade volumes, product categories, and regional competitiveness, offering insights into supply chains and market demand. This section also analyzes past and future pricing trends, helping businesses optimize strategies and enabling consumers to assess product value effectively.
Medical Device Testing Services Market: Company Profiles
The report identifies key players in the medical device testing services market through competitive landscape and company profiles, evaluating their offerings, financial performance, strategies, and market positioning. It includes a SWOT analysis of the top 3-5 companies, assessing strengths, weaknesses, opportunities, and threats. The competitive landscape highlights rankings, recent activities (mergers, acquisitions, partnerships, product launches), and regional footprints using the Ace matrix. Customization is available to meet client-specific needs.
Regional & Industry Footprint
This section details the geographic reach, sales networks, and market penetration of companies profiled in the medical device testing services report, showcasing their operations and distribution across regions. It analyzes the alignment of companies with specific industry verticals, highlighting the industries they serve and the scope of their products and services within those sectors.
Ace Matrix
This section categorizes companies into four distinct groups—Active, Cutting Edge, Innovator, and Emerging—based on their product and business strategies. The evaluation of product strategy focuses on aspects such as the range and depth of offerings, commitment to innovation, product functionalities, and scalability. Key elements like global reach, sector coverage, strategic acquisitions, and long-term growth plans are considered for business strategy. This analysis provides a detailed view of companies' position within the market and highlights their potential for future growth and development.
Research Methodology
The qualitative and quantitative insights for the medical device testing services market are derived through a multi-faceted research approach, combining input from subject matter experts, primary research, and secondary data sources. Primary research includes gathering critical information via face-to-face or telephonic interviews, surveys, questionnaires, and feedback from industry professionals, key opinion leaders (KOLs), and customers. Regular interviews with industry experts are conducted to deepen the analysis and reinforce the existing data, ensuring a robust and well-rounded market understanding.
Secondary research for this report was carried out by the Market Research Store team, drawing on a variety of authoritative sources, such as:
- Official company websites, annual reports, financial statements, investor presentations, and SEC filings
- Internal and external proprietary databases, as well as relevant patent and regulatory databases
- Government publications, national statistical databases, and industry-specific market reports
- Media coverage, including news articles, press releases, and webcasts about market participants
- Paid industry databases for detailed market insights
Market Research Store conducted in-depth consultations with various key opinion leaders in the industry, including senior executives from top companies and regional leaders from end-user organizations. This effort aimed to gather critical insights on factors such as the market share of dominant brands in specific countries and regions, along with pricing strategies for products and services.
To determine total sales data, the research team conducted primary interviews across multiple countries with influential stakeholders, including:
- Distributors
- Marketing, Brand, and Product Managers
- Procurement and Production Managers
- Sales and Regional Sales Managers, Country Managers
- Technical Specialists
- C-Level Executives
These subject matter experts, with their extensive industry experience, helped validate and refine the findings. For secondary research, data was sourced from a wide range of materials, including online resources, company annual reports, industry publications, research papers, association reports, and government websites. These various sources provide a comprehensive and well-rounded perspective on the market.
Frequently Asked Questions
Who are the leading players functioning in the global medical device testing services market growth?
Table Of Content
Table of Content 1 Medical Device Testing Services Market - Research Scope 1.1 Study Goals 1.2 Market Definition and Scope 1.3 Key Market Segments 1.4 Study and Forecasting Years 2 Medical Device Testing Services Market - Research Methodology 2.1 Methodology 2.2 Research Data Source 2.2.1 Secondary Data 2.2.2 Primary Data 2.2.3 Market Size Estimation 2.2.4 Legal Disclaimer 3 Medical Device Testing Services Market Forces 3.1 Global Medical Device Testing Services Market Size 3.2 Top Impacting Factors (PESTEL Analysis) 3.2.1 Political Factors 3.2.2 Economic Factors 3.2.3 Social Factors 3.2.4 Technological Factors 3.2.5 Environmental Factors 3.2.6 Legal Factors 3.3 Industry Trend Analysis 3.4 Industry Trends Under COVID-19 3.4.1 Risk Assessment on COVID-19 3.4.2 Assessment of the Overall Impact of COVID-19 on the Industry 3.4.3 Pre COVID-19 and Post COVID-19 Market Scenario 3.5 Industry Risk Assessment 4 Medical Device Testing Services Market - By Geography 4.1 Global Medical Device Testing Services Market Value and Market Share by Regions 4.1.1 Global Medical Device Testing Services Value ($) by Region (2015-2020) 4.1.2 Global Medical Device Testing Services Value Market Share by Regions (2015-2020) 4.2 Global Medical Device Testing Services Market Production and Market Share by Major Countries 4.2.1 Global Medical Device Testing Services Production by Major Countries (2015-2020) 4.2.2 Global Medical Device Testing Services Production Market Share by Major Countries (2015-2020) 4.3 Global Medical Device Testing Services Market Consumption and Market Share by Regions 4.3.1 Global Medical Device Testing Services Consumption by Regions (2015-2020) 4.3.2 Global Medical Device Testing Services Consumption Market Share by Regions (2015-2020) 5 Medical Device Testing Services Market - By Trade Statistics 5.1 Global Medical Device Testing Services Export and Import 5.2 United States Medical Device Testing Services Export and Import (2015-2020) 5.3 Europe Medical Device Testing Services Export and Import (2015-2020) 5.4 China Medical Device Testing Services Export and Import (2015-2020) 5.5 Japan Medical Device Testing Services Export and Import (2015-2020) 5.6 India Medical Device Testing Services Export and Import (2015-2020) 5.7 ... 6 Medical Device Testing Services Market - By Type 6.1 Global Medical Device Testing Services Production and Market Share by Types (2015-2020) 6.1.1 Global Medical Device Testing Services Production by Types (2015-2020) 6.1.2 Global Medical Device Testing Services Production Market Share by Types (2015-2020) 6.2 Global Medical Device Testing Services Value and Market Share by Types (2015-2020) 6.2.1 Global Medical Device Testing Services Value by Types (2015-2020) 6.2.2 Global Medical Device Testing Services Value Market Share by Types (2015-2020) 6.3 Global Medical Device Testing Services Production, Price and Growth Rate of Biocompatibility Tests (2015-2020) 6.4 Global Medical Device Testing Services Production, Price and Growth Rate of Chemistry Test (2015-2020) 6.5 Global Medical Device Testing Services Production, Price and Growth Rate of Microbiology & Sterility Testing (2015-2020) 6.6 Global Medical Device Testing Services Production, Price and Growth Rate of Package Validation (2015-2020) 7 Medical Device Testing Services Market - By Application 7.1 Global Medical Device Testing Services Consumption and Market Share by Applications (2015-2020) 7.1.1 Global Medical Device Testing Services Consumption by Applications (2015-2020) 7.1.2 Global Medical Device Testing Services Consumption Market Share by Applications (2015-2020) 7.2 Global Medical Device Testing Services Consumption and Growth Rate of Preclinical (2015-2020) 7.3 Global Medical Device Testing Services Consumption and Growth Rate of Clinical (2015-2020) 8 North America Medical Device Testing Services Market 8.1 North America Medical Device Testing Services Market Size 8.2 United States Medical Device Testing Services Market Size 8.3 Canada Medical Device Testing Services Market Size 8.4 Mexico Medical Device Testing Services Market Size 8.5 The Influence of COVID-19 on North America Market 9 Europe Medical Device Testing Services Market Analysis 9.1 Europe Medical Device Testing Services Market Size 9.2 Germany Medical Device Testing Services Market Size 9.3 United Kingdom Medical Device Testing Services Market Size 9.4 France Medical Device Testing Services Market Size 9.5 Italy Medical Device Testing Services Market Size 9.6 Spain Medical Device Testing Services Market Size 9.7 The Influence of COVID-19 on Europe Market 10 Asia-Pacific Medical Device Testing Services Market Analysis 10.1 Asia-Pacific Medical Device Testing Services Market Size 10.2 China Medical Device Testing Services Market Size 10.3 Japan Medical Device Testing Services Market Size 10.4 South Korea Medical Device Testing Services Market Size 10.5 Southeast Asia Medical Device Testing Services Market Size 10.6 India Medical Device Testing Services Market Size 10.7 The Influence of COVID-19 on Asia Pacific Market 11 Middle East and Africa Medical Device Testing Services Market Analysis 11.1 Middle East and Africa Medical Device Testing Services Market Size 11.2 Saudi Arabia Medical Device Testing Services Market Size 11.3 UAE Medical Device Testing Services Market Size 11.4 South Africa Medical Device Testing Services Market Size 11.5 The Influence of COVID-19 on Middle East and Africa Market 12 South America Medical Device Testing Services Market Analysis 12.1 South America Medical Device Testing Services Market Size 12.2 Brazil Medical Device Testing Services Market Size 12.3 The Influence of COVID-19 on South America Market 13 Company Profiles 13.1 NORTH AMERICAN SCIENCE ASSOCIATES INC. 13.1.1 NORTH AMERICAN SCIENCE ASSOCIATES INC. Basic Information 13.1.2 NORTH AMERICAN SCIENCE ASSOCIATES INC. Product Profiles, Application and Specification 13.1.3 NORTH AMERICAN SCIENCE ASSOCIATES INC. Medical Device Testing Services Market Performance (2015-2020) 13.2 Toxikon, Inc. 13.2.1 Toxikon, Inc. Basic Information 13.2.2 Toxikon, Inc. Product Profiles, Application and Specification 13.2.3 Toxikon, Inc. Medical Device Testing Services Market Performance (2015-2020) 13.3 Eurofins Scientific, LLC 13.3.1 Eurofins Scientific, LLC Basic Information 13.3.2 Eurofins Scientific, LLC Product Profiles, Application and Specification 13.3.3 Eurofins Scientific, LLC Medical Device Testing Services Market Performance (2015-2020) 13.4 WuXi AppTec 13.4.1 WuXi AppTec Basic Information 13.4.2 WuXi AppTec Product Profiles, Application and Specification 13.4.3 WuXi AppTec Medical Device Testing Services Market Performance (2015-2020) 13.5 Charles River Laboratories International Inc. 13.5.1 Charles River Laboratories International Inc. Basic Information 13.5.2 Charles River Laboratories International Inc. Product Profiles, Application and Specification 13.5.3 Charles River Laboratories International Inc. Medical Device Testing Services Market Performance (2015-2020) 13.6 Intertek Group Plc 13.6.1 Intertek Group Plc Basic Information 13.6.2 Intertek Group Plc Product Profiles, Application and Specification 13.6.3 Intertek Group Plc Medical Device Testing Services Market Performance (2015-2020) 13.7 SGS SA 13.7.1 SGS SA Basic Information 13.7.2 SGS SA Product Profiles, Application and Specification 13.7.3 SGS SA Medical Device Testing Services Market Performance (2015-2020) 13.8 Sterigenics International LLC 13.8.1 Sterigenics International LLC Basic Information 13.8.2 Sterigenics International LLC Product Profiles, Application and Specification 13.8.3 Sterigenics International LLC Medical Device Testing Services Market Performance (2015-2020) 13.9 Pace Analytical Services 13.9.1 Pace Analytical Services Basic Information 13.9.2 Pace Analytical Services Product Profiles, Application and Specification 13.9.3 Pace Analytical Services Medical Device Testing Services Market Performance (2015-2020) 14 Market Forecast - By Regions 14.1 North America Medical Device Testing Services Market Forecast (2020-2025) 14.2 Europe Medical Device Testing Services Market Forecast (2020-2025) 14.3 Asia-Pacific Medical Device Testing Services Market Forecast (2020-2025) 14.4 Middle East and Africa Medical Device Testing Services Market Forecast (2020-2025) 14.5 South America Medical Device Testing Services Market Forecast (2020-2025) 15 Market Forecast - By Type and Applications 15.1 Global Medical Device Testing Services Market Forecast by Types (2020-2025) 15.1.1 Global Medical Device Testing Services Market Forecast Production and Market Share by Types (2020-2025) 15.1.2 Global Medical Device Testing Services Market Forecast Value and Market Share by Types (2020-2025) 15.2 Global Medical Device Testing Services Market Forecast by Applications (2020-2025)
Inquiry For Buying
Medical Device Testing Services
Request Sample
Medical Device Testing Services
